Liquid And Lyophilised Product Vial Filling Line
- Vial Washing Machine
- Hot Air Sterilization & Depyrogenation Tunnel
- Monoblock Vial Liquid Filling & Stoppering
- Aluminium Cap Sealing Machine
Vial Powder Filling Line
- Vial Washing Machine
- Hot Air Sterilization & Depyrogenation Tunnel
- Powder Filling Machine
- Aluminium Cap Sealing Machine
Ampoule Filling Line & Combo Ampoule
- Ampoule & Vial Washing Machine
- Sterilization & Depyrogenation Tunnel
- Ampoule Filling & Sealing Machine
R & D Application Machine
Dry Syrup Filling Line
Rotary Filling Pumps For Liquids
PP Injection Stretch Blow Moulding & Filing Line(ISBM)
Camera Inspection Machine For Container
Revamping Of Filling Machine ⇓
Revamping Of Filling Machine


 This Multi head Unit is specially designed to be integrated with filling system, hence provides opportunity to upgrade the filling system with servo mechanism for precise filling without modifying existing area layout.
This Multi head Unit is specially designed to be integrated with filling system, hence provides opportunity to upgrade the filling system with servo mechanism for precise filling without modifying existing area layout.
- Solution to problems inherent to filling stations,aiming at wiping out issues related to product,viscosity,drop splash etc.
- Individual control of filling pumps.
- Tool less change over filling syringes.
- Precision of filling +/- 0.1%.
- Easily integrated to any make of existing filling machine.
- Available in various number of filling head depending upon existing filling machine speed.
- Upgrading of filling to have precise filling without any area modification.
- Trouble free system hence gives freedom for mechanical setting of fill volume. Fine setting can be done through touch screen.
- Better filling hence less wastage and more yield.
- No mechanical spare consumable.
Surface Disinfection using H2O2,Dosy Mist ⇓
DOSYMIST The New Generation of H2O2 Generation System
 Solidfog offers you Simple,Reliable,Economical and Easy to validate decontamination system with H2O2.
Solidfog offers you Simple,Reliable,Economical and Easy to validate decontamination system with H2O2.
- Simple: Compact design, reduced size, mobile and easy to handle (plug-and-play).
- Reliable: Calibration certificate of components, Siemens PLC, control and self-diagnose of working parameters. IQ/OQ complete documentation.
- Economical: High quality for a fair price, use of non-proprietary biocide (H2O2 12%), better and wider materials compatibility.
- Ease Of Validation: Great dispersion capacity (up to 200 m3 volume), very high efficiency (up to 6-log) and capable of monitoring and recording data for cycle validation.
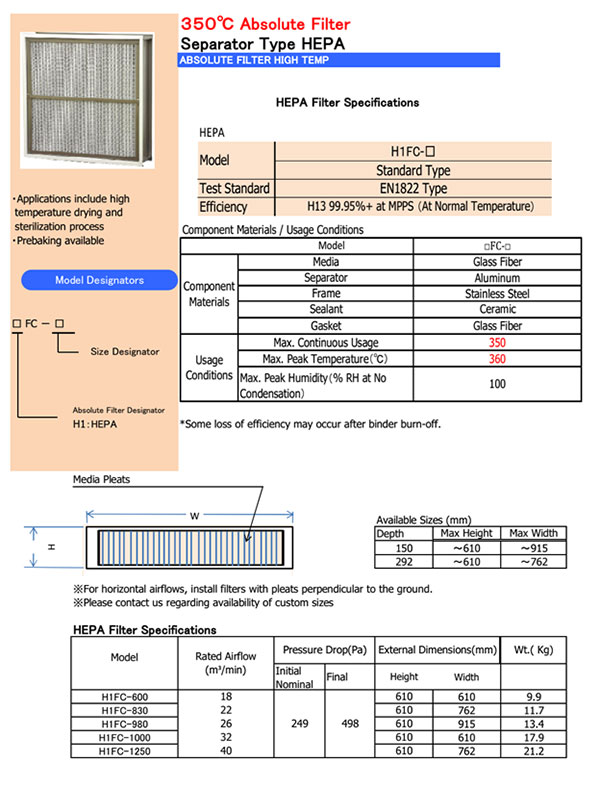
High Temperature HEPA H13 Filters for 350OC Application ⇓
High Temperature Filter Selection Guide
 Cambridge High Temperature Filters may contain either ceramic or silicone sealant. As silicone sealant filters may release silicone gas during usage, for sterilization,filling, film coating and other similar processes, ceramic sealant filters are recommended.
Cambridge High Temperature Filters may contain either ceramic or silicone sealant. As silicone sealant filters may release silicone gas during usage, for sterilization,filling, film coating and other similar processes, ceramic sealant filters are recommended.
Automatic Glove Leak Testing System ⇓
It's advanced. Its automatic. It's above everything you have used so far.
 Other Key Features
Other Key Features
- Manufacturing materials resistant to sanitizing agents and bio decontamination process.
- Automatic complete testing procedures.
- Parametric testing formulas.
- Integrated pneumatic circuit.
- Complete encrypted data/report storage and download via SD card.
- Testing time from 10 minutes, depending on both glove material and dimension.
- Travelling ergonomic docking station made of SS304 (Configuration for 4 & 8 testing units available).
- Low start-up costs.
- Saving production time in isolated filling line, stand alone isolator systems, and RABS Technology.
- Avoiding operator mistakes.
- Detecting small holes in the glove (> 50�m).
- Data consulting can be done outside the classified environment.
Automation
- Touchscreen PC control station.
- Dedicated Windows based software application for storage and report of the transmitted data available.
- Software development according to Good Automated Manufacturing Practice (GAMP 5).
- 21 CFR Part 11 Complaint.
- Operator friendly interface available different languages.
- VPN connection for remote access and assistance ,updates and maintenance available.
High Containment Isolator Systems. ⇓
Tema Sinergie Containment Isolators have been designed for handling high potent compounds and offer guaranteed very high levels of operator protection.
 Tema Sinergie Containment Isolators have been designed for handling high potent compounds and offer guaranteed very high levels of operator protection.
Operator safety is crucial because of the potential dangerous nature of the materials. Maximizing operator protection is the primary characteristic of a containment isolator, a perfect combination of design and manufacturing strategies assure a 100% OEL5 (=50ng /m3).
Tema Sinergie Containment Isolators have been designed for handling high potent compounds and offer guaranteed very high levels of operator protection.
Operator safety is crucial because of the potential dangerous nature of the materials. Maximizing operator protection is the primary characteristic of a containment isolator, a perfect combination of design and manufacturing strategies assure a 100% OEL5 (=50ng /m3).
Product Transfer, Manual Sampling, Weighing and Dispensing operations of Highly Potent Active Pharmaceutical Ingredients (HPAPI) are some of the default activities our Containment Isolators are designed for.
Restricted Access Barrier Systems (RABS). ⇓
Our equipment are designed to meet all existing requirements for aseptic processing. RABS provide protection by delivering a physical and aerodynamic barrier over a critical process zone with easier access to the process in the event a human intervention is required.
 Tema Sinergie develops different kinds of RABS.
Tema Sinergie develops different kinds of RABS.
- Open Passive RABS: barrier system which utilizes existing cleanroom overhead air supply systems to deliver HEPA filtered air over a critical process before returning air back into the clean room.
- Open Active RABS: barrier system which has an integrated ventilation system to supply HEPA filtered air over a critical process before returning air back into the clean room.
- Closed RABS: a positive pressure system which has an integrated ventilation system to supply HEPA filtered air over a critical process, which can pass through return filters before being recirculated. Closed RABS can also be integrated with Bio-decontamination system to provide with a CGMP class A/ISO 5 environment.
Aseptic Processing Isolator Systems ⇓
Aseptic Processing Isolator Systems designed for QC Labs and pharmaceutical production.
 The high level of aseptic conditions achievable makes these isolators the perfect flexible equipment for different applications, such as Sterility Testing, Aseptic Dispensing & Sampling, and Aseptic Product Transfer. Specific applications for Beta emitters available.
The high level of aseptic conditions achievable makes these isolators the perfect flexible equipment for different applications, such as Sterility Testing, Aseptic Dispensing & Sampling, and Aseptic Product Transfer. Specific applications for Beta emitters available.
Recirculating Unidirectional Air Flow with return filters, and integrated Bio-decontamination system to allow 6-log bacterial reduction. Rapid Gas Chamber (RGC) with integrated VPHP technology for a fastest and safest bio decontamination transfer process (Tmax = 25'full aeration, VHP concentration = 1ppm).
Modular based configurations or custom design to fit specific user requirements.
Active Bio-decon Pass Through Chambers ⇓
Active Bio-decon Pass Through Chambers enable aseptic transfer of material in to cleanrooms.

- The materials are sterilized in the Pass-Through Chamber by means of integrated Bio-decontamination system.
- The integrated ventilation system of the chamber guarantees a pressure cascade between the adjoining rooms and the chamber itself.
- This cascade preserves the asepsis of the materials after the termination of bio-decontamination process prior to extraction into the cleanroom.
- Walk-in models available. Custom design to fit individual applications.
Precision Dispensing & Metering Pumps ⇓

Design and manufacture of
- Precision low-volume.
- Positive displacement.
- Liquid dispensing.
- Metering systems.
IVEK metering systems can deliver continuous flows from less then 100ul/minute to 3.5 litres/minute.
Diagnostic companies use our pumps for metering and dispensing reagents.
- Onto nitrocellulose membranes.
- Into micro titer plates.
- On bio-sensor cards.
- For high throughput screening.
- Diagnostic test strips.
Pharmaceutical companies have equipmentto fill small bottles with products like.
- Insulin
- Radioactive reagents.
- Coat stents.
- Siliconizing syringes(we coat more than 60% of all syringes manufactured for medical applications.
- Dose radioactive pharmaceuticals.
Food companies have systems that can add:
- Flavourings.
- Vitamins solutions.
- Sodium solutions
Chemical plants use our metering systems
- To precisely add acid or base solutions into mixing reactors..
- To use systems for replenishing liquid metals in plating processes.
- Filling acid into batteries.
Customised Automation ⇓
Customised Automation

- PBL is the ideal partner for customized solutions applied to primary and secondary packaging. In a collaborative approach, our experts offer flexibility, innovation, technical expertise, and value on your investment.
Customers looking for complex and customized software and hardware solutions can find an ideal partner at PBL. Our expertise in Artificial Intelligence and Deep Learning are offered to companies in need of dedicated industrial solutions based on innovative and efficient technologies.
Ready to Use ( RTU ) Containers Syringes/Vials/ Cartridges processing line consisting of Washer, Siliconisation, Depyrogenation, Capping with Luvers/Needle Assembly &Renesting into Nest is one of the best solutions of Automation offered by PBL.

Production : 12,000 Syringes/ Vials per Hour
We also work for Food Industry such as Coffee Cups filling Lines, Beverages & wherever Automation is needed.


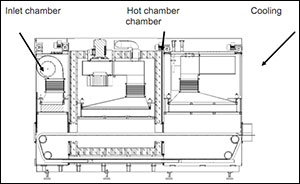
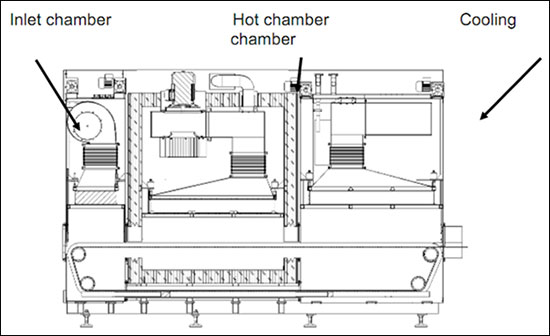
 The hot Air Sterilization & Depyrogenation laminar air flow tunnels sterilize and Depyrogenates glass vials. Unidirectional Laminar air flow preheats, Depyrogenates and cools the vials in an ISO 5 environment attaining a 3 log bio-burden reduction of Endotoxins.
The hot Air Sterilization & Depyrogenation laminar air flow tunnels sterilize and Depyrogenates glass vials. Unidirectional Laminar air flow preheats, Depyrogenates and cools the vials in an ISO 5 environment attaining a 3 log bio-burden reduction of Endotoxins.




 The AFM series machines are designed to process both closed and open ampoules. Developed to meet current pharmaceutical industry requirements, and to address the needs of the enduser, they are suitable both for liquid parenterals and products that must undergo the lyophilisation process.Their extremely slim design reduces expensive classified area space requirements, though within a conventional clean room, or with other containment systems such as RABS or isolators. It also allows different layout configurations and center of the room or wall installation.
The AFM series machines are designed to process both closed and open ampoules. Developed to meet current pharmaceutical industry requirements, and to address the needs of the enduser, they are suitable both for liquid parenterals and products that must undergo the lyophilisation process.Their extremely slim design reduces expensive classified area space requirements, though within a conventional clean room, or with other containment systems such as RABS or isolators. It also allows different layout configurations and center of the room or wall installation.
 The SYFILL series of fiiling and stoppering machines have been specially designed to process already sterile nested containers, such as syringes, cartridges, and vials.Developed to meet current pharmaceutical industry standards and requirements, the SYFILL Series has a particularly compact design that reeduces the space taken in sterile room.
The SYFILL series of fiiling and stoppering machines have been specially designed to process already sterile nested containers, such as syringes, cartridges, and vials.Developed to meet current pharmaceutical industry standards and requirements, the SYFILL Series has a particularly compact design that reeduces the space taken in sterile room.
 This is single head filling system designed to fill from 0.001ml to 150ml with different set of syringes.It is designed to adjust pump volume by weight. It is provided with Quick fitting of the pump with the patented system KLYX. It is 21CFR part 11 Compliant.
This is single head filling system designed to fill from 0.001ml to 150ml with different set of syringes.It is designed to adjust pump volume by weight. It is provided with Quick fitting of the pump with the patented system KLYX. It is 21CFR part 11 Compliant. The M & O Perry Powder filler are designed using the ACCOFIL Technology, a patented technology in 1948.Thanks to our Experience of more than 60 years, which makes our Machines one of the best in World. Macro dose Powder Filling machines accurately dose large fills of powder into Glass and plastic Bottles with Principal of ACCOFIL. An optional capping turret applies ROPP, snap-on, screw type or tamper proof caps onto bottles. Typical applications includes ORAL ANTIBIOTICS AND BABY POWDER into glass and plastic bottles and ASPRIN IN POUCHES.
The M & O Perry Powder filler are designed using the ACCOFIL Technology, a patented technology in 1948.Thanks to our Experience of more than 60 years, which makes our Machines one of the best in World. Macro dose Powder Filling machines accurately dose large fills of powder into Glass and plastic Bottles with Principal of ACCOFIL. An optional capping turret applies ROPP, snap-on, screw type or tamper proof caps onto bottles. Typical applications includes ORAL ANTIBIOTICS AND BABY POWDER into glass and plastic bottles and ASPRIN IN POUCHES.


 SPC, a 100 Years old company, is a manufacturer of Tailor Made devices such as Rotary Piston Pumps, Needles fo⇓r Filling Machines & Washing Machines.The word SPC is synonymous to Your precision partner for liquid filling The various industry we serve are pharmaceuticals, machinery manufacturer of sterile equipments (oem s), cosmetics, biotech & the food industry.
SPC, a 100 Years old company, is a manufacturer of Tailor Made devices such as Rotary Piston Pumps, Needles fo⇓r Filling Machines & Washing Machines.The word SPC is synonymous to Your precision partner for liquid filling The various industry we serve are pharmaceuticals, machinery manufacturer of sterile equipments (oem s), cosmetics, biotech & the food industry.




 This Machine can process Ampoules, Vials and Cartridges filled with sterile products.These are fully automated inspection machines represents the ideal solution to comply GMP requirements and to ensure that injectable products are essentially free of contamination.In standard configuration each containers are inspected at least three times for particles. If deemed necessary a fourth station can also be installed.
This Machine can process Ampoules, Vials and Cartridges filled with sterile products.These are fully automated inspection machines represents the ideal solution to comply GMP requirements and to ensure that injectable products are essentially free of contamination.In standard configuration each containers are inspected at least three times for particles. If deemed necessary a fourth station can also be installed. This Machine can process Ampoules, Vials and Cartridges filled with sterile products.These are fully automated inspection machines represents the ideal solution to comply GMP requirements and to ensure that injectable products are essentially free of contamination.In standard configuration each containers are inspected at least three times for particles. If deemed necessary a fourth station can also be installed.
This Machine can process Ampoules, Vials and Cartridges filled with sterile products.These are fully automated inspection machines represents the ideal solution to comply GMP requirements and to ensure that injectable products are essentially free of contamination.In standard configuration each containers are inspected at least three times for particles. If deemed necessary a fourth station can also be installed. When processing containers immediately after autoclaving, there might be the need to eliminate residual water or moisture and so a special drying module can be added to the machine. Containers are externally dried to finally undergo leak testing and or particulate and cosmetic inspection for drying module on request.
When processing containers immediately after autoclaving, there might be the need to eliminate residual water or moisture and so a special drying module can be added to the machine. Containers are externally dried to finally undergo leak testing and or particulate and cosmetic inspection for drying module on request.